- Blog
- Seavus project viewer cost
- Infinity kingdom immortal tier list reddit
- Adidas daily 2
- Pulsar lost colony key
- Coromon app
- Ram pressure meteor
- Ark survival evolved phiomia
- Lattice energy equation
- Ilift apple app android
- Turbo speed reader download
- Automailer storyboard text messages
- Call of duty modern warfare 2 bot
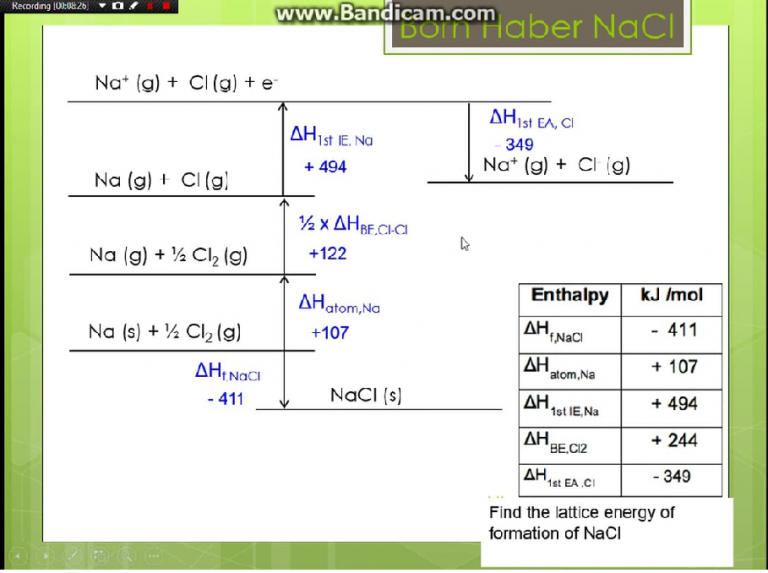
Where: G U denotes the molar lattice energy.
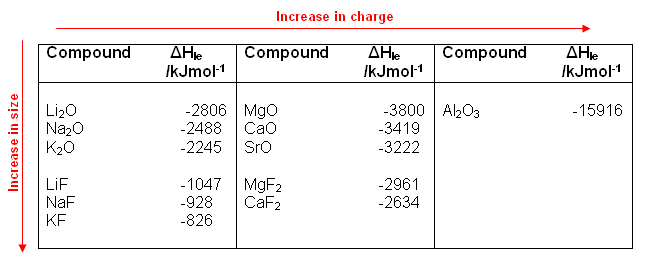
The following equation can be used to represent the molar lattice energy of an ionic crystal in terms of molar lattice enthalpy, pressure, and volume change: G U G H pV m. Lattice energy cannot be determined experimentally due to the difficulty in isolating gaseous ions. Abstract-Evaluation of internal energy and the inter-atomic or ionic interactions in a crystal lattice usually requires precise calculation of lattice sums. Although lattice energy cannot be measured experimentally, it can be calculated or estimated using electrostatics or the Born-Haber cycle.

NaCl Crystalline LatticeSodium ions (Na +) and chloride(Cl -) ions, depicted in purple and green respectively, alternate in the crystal lattice of solid NaCl. Calculation of the lattice energy and the energy gap of the magnetic semiconductor MnGa2 Se4 using Hartree-Fock and density functional theory methods. A high negative value of lattice energy indicates that the formation of NaCl is highly favored. The negative sign of the energy is indicative of an exothermic reaction.Īlternatively, lattice energy can be thought of as the energy required to separate a mole of an ionic solid into the gaseous form of its ions (that is, the reverse of the reaction shown above). While, known formulae like the Kapustinskii equation predict lattice energies for alkali halides that are better in agreement with experimental results (within. The value of its lattice energy is -787 kJ/mol.
